Yay, Lou was accepted into an Immunotherapy trial today. A tiny win in our cancer war.
The Good
The study might work. Fingers and toes crossed. Pray to your gods.
If the study doesn’t work for us, the knowledge will help people with advanced cancer in the future. That’s a good thing.
The Bad
A very long list of side effects, several pages. But the doctors like to cover their arse and we’ve seen the worst, so bring it.
The Ugly
We may not receive any benefits from this research and it is possible Lou’s condition may become worse during the study. We accept this and are already on the case of alternative options.
Some detail about this Immunotherapy Trial – Only if you are interested.
These studies are run by the big drug companies, in our case, it’s Pfizer, Inc. They pay an organisation to conduct the studies. Luckily for us, one of the locations is Macquarie University, where we will join the study. I guess the big win for the drug company is if they succeed they have the patented right to sell the drug commercially.
What is the purpose of this specific research?
The purpose of this research study is to learn about the effects of the study drugs, avelumab and talazoparib when given as a combination treatment for Lou’s triple negative breast cancer. Avelumab and talazoparib have never been given together as a combination treatment, so the research study will also find the best dose of talazoparib to be given in combination with avelumab. Avelumab and talazoparib are both investigational drugs. (An investigational drug is one that is not approved for sale by regulatory authorities.)
A little bit more about the drug coctail, Lou loves a cocktail
Avelumab is a monoclonal antibody, a type of protein that recognises and attaches to other proteins. Avelumab specifically recognises and attaches to a protein called PD-L1. PD-L1 is found on the surface of some cancer cells, where it acts to protect those cells from being attacked by your immune system (the part of your body that fights infection but which is also involved in fighting cancer). When avelumab attaches to PD-L1 it stops PD-L1 from working and so allows your immune system to recognise and kill your cancer cells.
Talazoparib is a drug that stops the activity of a protein called poly (adenosine diphosphate [ADP]-ribose) polymerase or PARP. PARP is involved in repairing damage to the DNA within your cells. DNA is the set of instructions found within all of your cells that tells them how to behave. Your DNA is damaged all the time by things around you in the environment, and is repaired by several different methods, one of which uses PARP. When PARP is turned off by talazoparib in your normal cells, other methods can still work to repair damage to DNA. However, in some cancer cells, these other methods are broken and cannot be used. When PARP is turned off by talazoparib in these cancer cells, DNA damage cannot be repaired and leads to the death of the cancer cells.
By giving avelumab and talazoparib as a combination treatment in this research study, they are evaluating if the study drugs work together to have a greater effect than if each study drug was given by itself.
A bit to take in there I know. If you don’t understand it all you can ask our little scientist Noah.
A total of about 300 patients are expected to participate in the study. The study is being done at approximately 50 different research centres globally. It is expected that approximately 6-10 patients will be enrolled at each research site.
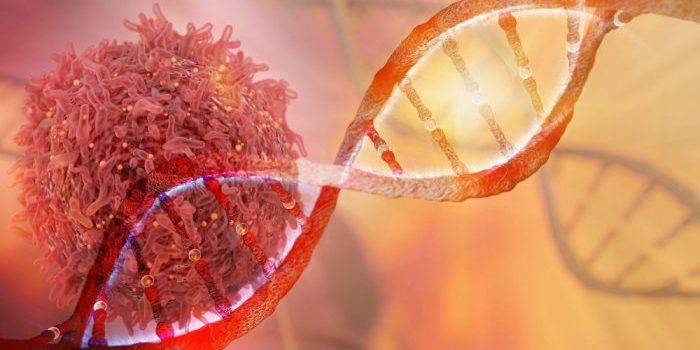
One in 300 and then one of 5-10 sound like strong stats
Being selected is amazing – keep fighting team – we are with you x x
Wow, sounds so full on! Fingers, toes and everything crossed that these drugs kill those cancer cells! Huge love and strength to you all xxx